Initial Theme: Electrochemical Materials for Energy Storage
Developing Next-Generation Electrolytes
Every advancement toward higher-energy or longer-lasting batteries demands a parallel advancement in electrolytes. The Leverick Lab studies non-equilibrium transport, ion-hopping mechanisms, and thermodynamics of complex electrolyte formulations to enable molecular-level design.
Li⁺ Hopping as the Key to Conductivity
Li⁺ conduction in most electrolytes is limited by slow, solvent-coupled diffusion, so we use model systems to isolate and study pure hopping mechanisms, revealing how molecular structure and defects control the barriers that govern conductivity across solid, polymer, and liquid systems
Decoding the Electrode-Electrolyte Interface
The true magic of a battery lies at the interface between the electrode and electrolyte, where chemical energy is converted into electrical energy. Yet despite its importance, many molecular mysteries remain that limit our ability to fully control battery reactions.
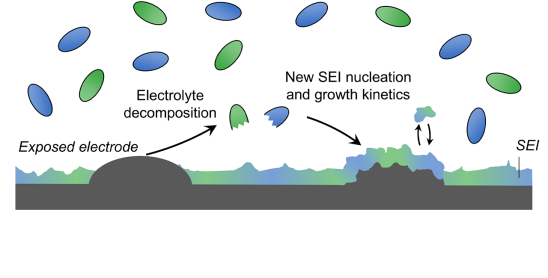
Beyond SEI Composition: Understanding How It Forms
Beyond just determining what the solid–electrolyte interphase (“SEI” - a protective layer on electrode surfaces) is made of, we use advanced characterization methods to reveal how this interphase nucleates and grows and how electrolyte composition can control it.
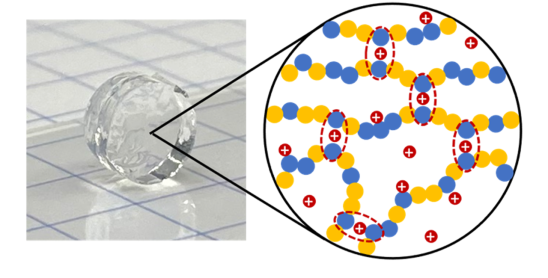
Ionogel Electrolytes via Polymer Self-Assembly
Room temperature molten salts known as ionic liquids offer exceptional nonvolatility and nonflammability for developing safer battery electrolytes. Supporting these materials with self-assembled polymer networks that leverage noncovalent charge-based interactions, free-standing ionogels that eliminate liquid leakage concerns and may be recycled can be realized.
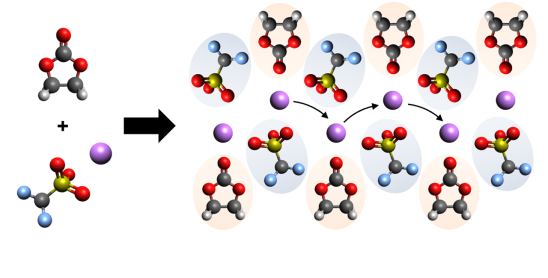
Alkali Metal Cation-mediated Coulombic Cross-links
We are investigating the ability of both Li+ and Na+ cations to spontaneously form robust, noncovalently cross-linked zwitterionic polymer networks inside of ionic liquid electrolytes. Within these safer electrolyte materials of the future, the alkali metal cations take on dual roles, acting both as gelator and as mobile charge carrier.